Commercial Step Therapy May Include Steps Beyond FDA Label
Summary
Across three therapeutic areas, an Avalere Health analysis identified commercial plan coverage policies with more steps than indicated by drugs’ FDA labels.Background
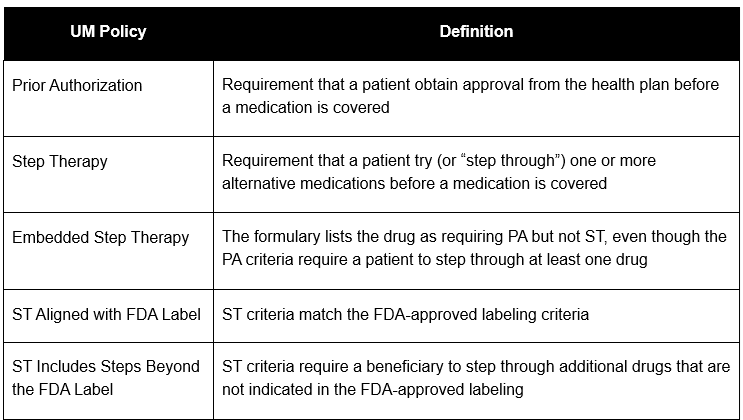
Step therapy (ST) is a utilization management (UM) practice that “establishes a sequence of covered prescription drugs for a given medical condition.” The ST process requires a plan enrollee to try or “step through” one or more alternative medications for a period (e.g., three months) before a medication is covered by the plan. Commercial plans may note on their formularies or in formulary lookup tools that a drug requires ST (“labeled ST”) or may include ST within prior authorization (PA) requirements (“embedded ST”). Because embedded ST is not labeled explicitly on formularies, individuals enrolling in a plan or looking up their drug coverage information would need to reference more detailed plan coverage policies to learn that a drug with embedded ST has such a requirement.
In some instances, plans may apply ST that requires an enrollee to step through additional drugs beyond what is indicated in Food & Drug Administration (FDA) approved labeling.¹
Avalere Health analyzed UM patterns in commercial plan pharmacy benefit coverage for branded, single-source drugs indicated to treat three conditions: psoriatic arthritis (PsA), multiple sclerosis (MS), and chronic myelogenous leukemia (CML) (UM policy definitions are provided in Table 1). Avalere Health conducted a previous analysis of the use of step therapy for specific drug indications across Medicare Part D.
Table 1: Definitions of UM Policies Included in the Analysis
Findings: Coverage and Use of Utilization Management
Avalere Health evaluated coverage for PsA, MS, and CML drugs on the pharmacy benefit in 2023 plans. Coverage and UM for each drug varied by plan, plan type, and benefit offering.
Commercial plans provided coverage 87% of the time for PsA and CML drugs, while only providing coverage 77% of the time for MS drugs.
Across commercial plans, UM strategies across the studied therapeutic areas vary widely. The following statistics show the portion of covered enrollees subject to a particular UM requirement (Figure 1).
- 90% of the time, covered PsA drugs are subject to UM
- 79% of the time, covered MS drugs are subject to UM
- 82% of the time, covered CML drugs are subject to UM
Commercial plans may require ST in one of two ways; either labeled as ST on the formulary or embedded within a PA requirement. Across PsA, MS, and CML drugs, commercial plans use embedded ST more frequently than labeled ST on the formulary.
- For PsA drugs, ST is embedded in the PA requirement 59% of the time, while it is labeled 11% of the time
- For MS drugs, ST is embedded 32% of the time and labeled 8% of the time
- For CML drugs, ST is embedded 51% of the time and labeled 12% of the time
ST may also be used in alignment with the FDA-approved labeling or extended further to include steps beyond the FDA label. For PsA, MS, and CML drugs subject to ST, plans may require enrollees to step through medications beyond those indicated by the FDA label.
- For PsA drugs, ST beyond the FDA-approved labeling is required 63% of the time
- Plans apply ST beyond the FDA label less frequently for MS and CML—37% and 39% of the time, respectively
Figure 1. Commercial Plan Utilization Management by Conditions and Benefit Type; Single-Source Brand Drugs, as a Percent of Lives with Drug Coverage, 2023
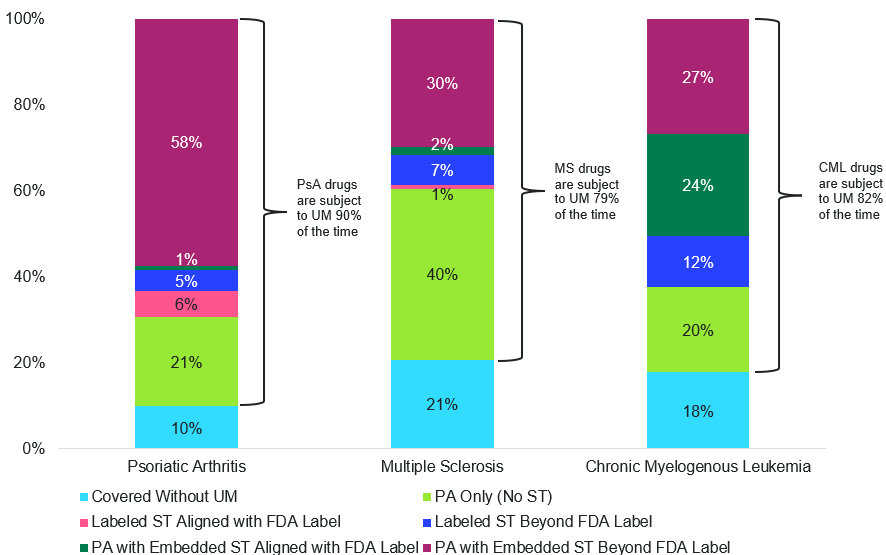
Note: Totals may not sum to 100% due to rounding.
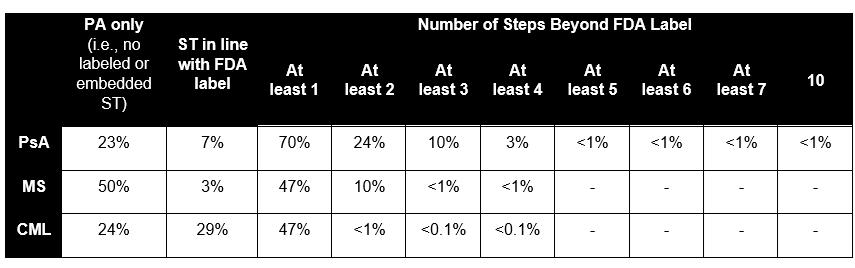
The number of steps required beyond the FDA-approved labeling varies widely by plan and drug. Across the drugs analyzed, the number of additional steps ranged from one to 10. When plans covered the 13 PsA drugs and required UM, they most frequently (60% of the time) applied one or two steps beyond the FDA label. A small portion of enrollees may be required to step through up to 10 therapies (beyond those specified on the FDA label) for three of the 13 PsA drugs (Table 2).
For MS drugs, one to two steps beyond the FDA-approved labeling is the most common ST policy; enrollees seeking two of the 11 MS drugs may face up to four steps beyond the label. For CML drugs with steps required beyond the FDA label, three of the four drugs analyzed require one additional step.
Table 2. Use of Step Therapy for Pharmacy-Benefit Drugs; Single-Source Brand Drugs, as a Percent of Lives with UM, 2023
Key Considerations
These findings offer insights into the ST landscape for individuals with chronic conditions. As other researchers have found, ST can delay patients’ access to needed medicines and impact patient care. The overlap of multiple steps in an ST protocol and the use of embedded ST may make the extent of ST requirements less obvious to individuals enrolling in coverage.
Although most states have legislation governing ST protocols and transparency, these laws do not address the aspects of ST considered in this analysis. Some laws outline exceptions and appeals processes for ST, but exact processes may vary by insurer. Federal policymakers, including those in the first Trump administration, have focused on policies requiring data transparency from health insurance plans that did not directly relate to ST.
Looking ahead, policymakers and employers selecting coverage for their employees could consider whether there are meaningful guardrails requiring oversight and transparency of ST protocols to ensure that individuals enrolling in a plan understand the ST requirements on a needed medication.
Methodology
Avalere Health evaluated the 2023 coverage and restrictions policies for branded, single-source drugs within three conditions (chronic myelogenous leukemia, multiple sclerosis, and psoriatic arthritis) on the pharmacy benefits for commercial plans. Commercial plans in this analysis include exchange plans, employer sponsored plans, federal employee health plans, union plans and municipal health plans. Avalere Health partners with Clarivate to obtain formulary and restrictions data. Restrictions data only includes policies from the top 250 plan sponsors, based on enrollment.
For each drug, Avalere Health evaluated whether the plan required ST (either explicitly listed as a step therapy or embedded in PA criteria), which drugs were listed as step edits, whether the step edit was aligned with the FDA-approved labeling or included more steps than the FDA label, and the number of step edits required for each condition. The UM criteria for each drug was compared to the FDA label. If the ST criteria matched the FDA label criteria, then the ST criteria was categorized as aligning with the FDA label. If the ST criteria required step edits beyond what the FDA label indicated (e.g., FDA label indicated no step edits, but ST criteria required a step edit), then the ST criteria was categorized as ST including steps beyond than the FDA label. Avalere Health did not evaluate how the UM criteria conform to clinical guidelines.
Dive Deeper
To learn more about the impact and prevalence of UM in the commercial market, connect with an Avalere Health expert today.
¹ The purpose of the FDA drug label is to provide health care providers with a summary of the safety and efficacy of a drug. FDA labels do not place specific limitations on the number of steps required for a prescription drug.






