A large life sciences manufacturer anticipated that some of its products may be selected for drug price negotiations under the Inflation Reduction Act (IRA). It sought to create an evidence strategy to articulate the value of its products during negotiations and thus needed to predict how CMS might weigh evidence and evaluate results. We helped the client anticipate CMS’s approach to evaluating evidence packages for products during the negotiation process, and develop strategies to strengthen those packages, to ensure that negotiated maximum fair prices (MFPs) accurately reflect the value of the client’s products.
Featured Insight
Trusted Partnership
We partner closely with clients to ensure their IRA strategy is tailored to their industry position, needs, and goals, helping clients make informed decisions and achieve long-term success.
Insight
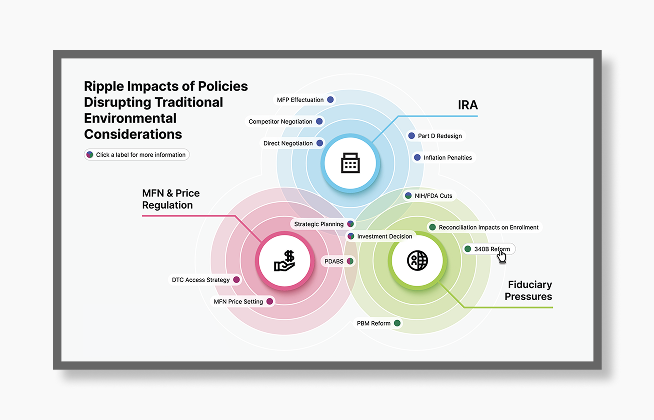
Planning for New Environmental Considerations
IRA Medicare provisions, increased focus on price regulations including Most-Favored Nation pricing, and broad fiduciary pressures have ripple effects across the healthcare ecosystem.
How Can We Help?
Engage an Expert Today.
Engage an Expert Today.


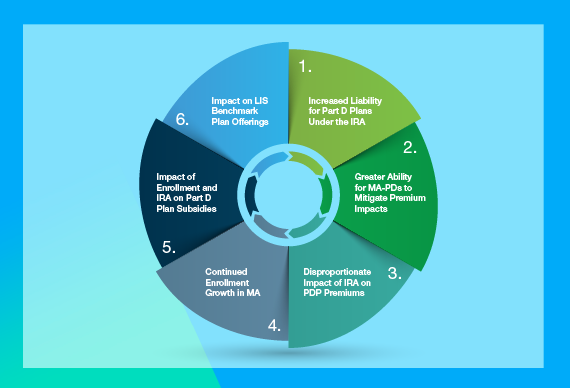
IRA, MFN, and Ongoing Fiduciary Pressures Creates Ripple Effect Across the Healthcare Ecosystem
IRA Medicare provisions, increased focus on price regulations including Most-Favored Nation pricing, and broad fiduciary pressures have ripple effects across the healthcare ecosystem.

Stakeholder Considerations for IPAY 2028 Guidance
Draft guidance for IPAY 2028 Medicare drug price negotiation includes the first-time inclusion of Part B drugs, refinement to MFP effectuation, and considerations around what qualifies as a single-source drug.
Stakeholder Considerations for MFP Effectuation in Part B
Several pathways exist to effectuate the MFP for Part B negotiation with benefits and drawbacks for stakeholders across the drug supply chain.

Early Enrollment Data Indicates More Beneficiaries Could Benefit from MPPP
Avalere Health’s analysis of early MPPP enrollment shows that only a small portion of beneficiaries who are likely to benefit from the program have enrolled.

Impact of MFP Effectuation on Pharmacies and Beneficiaries
A large share (30%) of beneficiaries access IPAYs 2026 or 2027 negotiated drugs through independent or franchise pharmacies.

Updated: Key Considerations for MFP Effectuation and the 340B Rebate Model
With less than a year until the first MFPs take effect, stakeholder concerns remain, especially as it relates to 340B duplicate discount risk.

White Paper: Valuing Multi-Use Drugs in Medicare Negotiation
Alternative approaches for weighting multiple uses of a product when setting maximum fair prices may better balance patient needs and development incentives.

IPAY 2027 Negotiated Drugs Expand Impact on Beneficiaries
The addition of 15 drugs selected for IPAY 2027 negotiation expand the impact maximum fair prices may have on key therapeutic areas and millions of beneficiaries.

Estimating the Spillover Impact of IRA Part B Negotiation
A survey finds that reimbursement contracts outside Medicare FFS still reflect Medicare metrics, raising questions about broader Part B negotiation effects.

CMS Announces Next 15 Drugs Selected for IRA Negotiation
The next cohort of drugs includes additional cancer and chronic disease therapies, as well as products that were therapeutic alternatives for IPAY 2026 drugs.
 Please wait ...
Please wait ...