New Clinical Trial Requirements Could Affect Vaccine Development
Summary
Reports that all new vaccines could require placebo-controlled testing are raising stakeholder concerns about the future of vaccine development, approval, and access.In a statement to the Washington Post on May 1, a spokesperson from the US Department of Health and Human Services (HHS) noted that “all new vaccines will undergo safety testing in placebo-controlled trials prior to licensure.” The recent appointment of Dr. Vinay Prasad to lead the Food and Drug Administration’s (FDA) Center for Biologics Evaluation and Research may signal potential shifts in regulatory emphasis, although no formal policy has yet been announced. Prior to his appointment, Dr. Prasad was a vocal critic of Operation Warp Speed, which was the US government’s interagency effort to expedite the development of COVID-19 vaccines and therapeutics.
Per current regulatory procedures, first-in-class vaccines (i.e., new vaccine candidates that are designed to prevent diseases for which no alterative and effective treatments or preventions exist) undergo placebo-controlled testing. As the FDA has yet to publish a formal announcement detailing this proposed requirement, it is unclear what “new vaccines” might be subject to it (though the HHS spokesperson’s statement indicated seasonal influenza vaccines may be exempt). Below, we detail the current procedures for studying vaccine safety and effectiveness and discuss potential impacts in the context of two types of vaccines most likely to be affected (seasonal and next-generation vaccines), should this requirement be implemented.
Current Vaccine Study Procedures
Generally, FDA-approved immunizations require extensive, multi-phase clinical trials before they are approved for use in the United States. These trials often compare a vaccine candidate to a placebo; specifically, trials of first-in-class vaccine candidates use a placebo comparator to establish non-rare safety events and side effect profiles. For example, the first COVID-19 vaccines were compared to placebo across all phases of human study.
Next-generation vaccines, which we define here as those that improve upon available products, are studied in comparison to the current standard of care. For example, a higher valent candidate in an existing vaccine class (e.g., pneumococcal, meningococcal) would be studied compared to available vaccines to understand the safety, efficacy, and immunogenicity of the candidate. These trials are designed to measure incremental improvements in vaccine efficacy and be more sensitive to rare safety events than a placebo-controlled trial.
Updated versions of licensed seasonal respiratory vaccines often do not require additional clinical data, although live attenuated, intranasally administered influenza vaccines are studied in small samples (approximately 300 adults) as part of their supplemental Biologics License Application to verify strain attenuation. Historically, updated COVID-19 vaccines have been developed over a similar timeline and undergone similar regulatory approval processes, with some year-to-year variation given differences in manufacturing timeline needs for different platform technologies (e.g., mRNA). For example, in 2024, the Vaccines and Related Biological Products Advisory Committee selected new vaccine strains in June in anticipation of vaccine availability in early fall.
Potential Scenarios and Stakeholder Concerns
The types of immunizations that could be most affected by changes in clinical study requirements are (1) seasonal respiratory vaccines (e.g., COVID-19), which are updated versions of existing products, and (2) next-generation vaccines, which aim to improve upon existing products.
Seasonal Respiratory Vaccines: Seasonal respiratory vaccines are updated annually (to reflect the latest surveillance data on dominant virus strains), so the potential impact of regulatory changes may be most clearly understood in these products. While updated influenza vaccines are developed over the approximately six months between when the FDA determines US strain composition and the beginning of the annual respiratory virus season, the development of updated COVID-19 vaccines has historically taken place over a much shorter timeline. As these COVID-19 vaccines are updated versions of already-approved products, the FDA does not require additional clinical study. This allows the vaccines to be quickly deployed throughout the US.
Stakeholders have expressed concern that, if implemented, a requirement for placebo-controlled trials in seasonal vaccine updates could extend development timelines and affect availability. Phase III trials, which test vaccine safety and efficacy, can take between six and nine months on an accelerated timeline. Additional time required for manufacturing, lot testing, and lot release can further extend the development timeline into and beyond respiratory virus season, limiting timely vaccine availability.
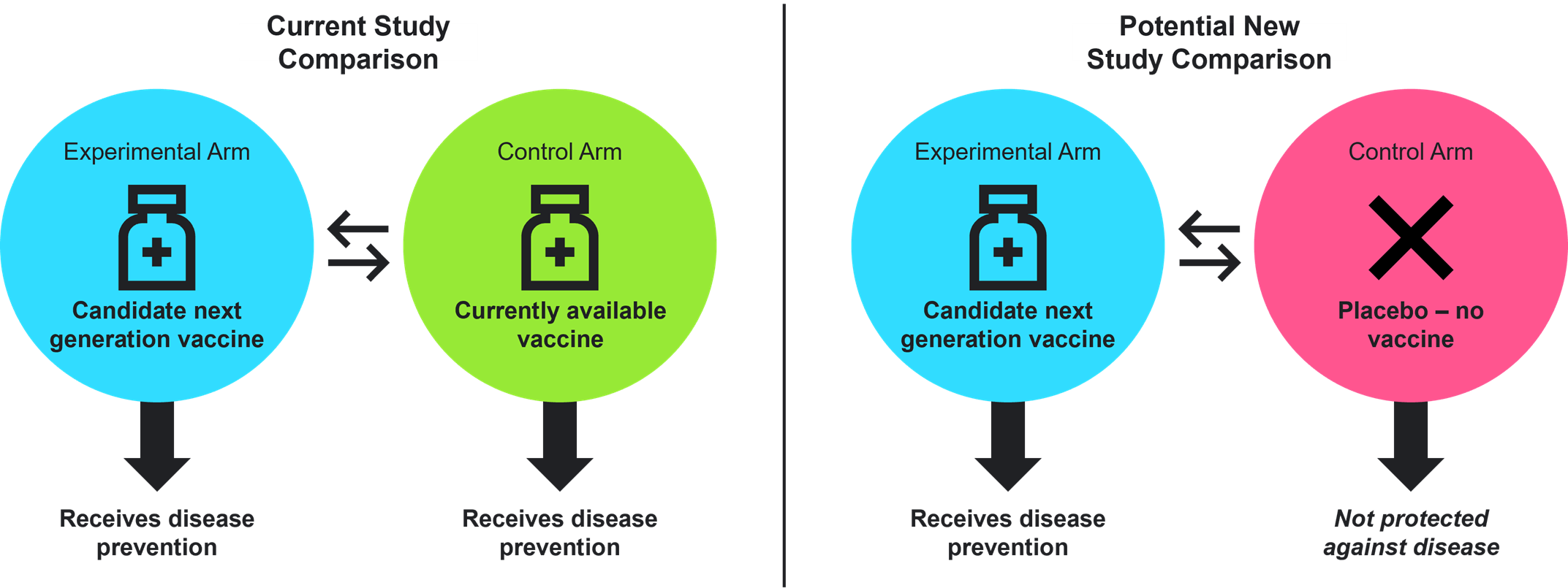
Next-Generation Vaccines: Next–generation vaccines are developed to improve upon currently available products. These vaccines are studied against their FDA-approved predecessors to determine the magnitude of that improvement. In these trials, participants in both the experimental arm and the control arm receive an immunization against the disease target in question.
If placebo-controlled trials are required for approval of next-generation vaccines, some stakeholders have raised ethical concerns, given that participants randomized to a trial’s control arm would receive a placebo. These participants would thus be potentially exposed to a vaccine-preventable disease target despite the availability of an FDA-approved vaccine (Figure 1).
Figure 1. Current vs. Potential New Next-Generation Vaccine Study Comparisons
Looking Ahead
It is unclear how and when the FDA would issue updated guidance. Stakeholders should continue to monitor the evolving debate and proactively consider how future changes could affect vaccine development and regulatory planning.
As the federal and state vaccine policy landscape and funding continues to change, stakeholders should continue to remain adaptable to shifts in requirements from federal agencies, including the FDA. To learn how Avalere Health’s Vaccines Team can help you prepare flexible vaccine policy engagement and evidence generation strategies in the face of uncertainty, connect with us.