Update: Health Plans’ Perceptions of PDABs and UPLs
Summary
In interviews and a survey, health plans communicate concerns with UPLs’ potential impact on patient access, pharmacists, providers, and their own financial positions.Building on research released in 2024, Avalere interviewed and surveyed health plan employees to assess how health plans perceive Prescription Drug Affordability Boards’ (PDAB) upper payment limits (UPLs) and impacts on patients, providers, and other stakeholders.
Key Findings
Respondents anticipate that UPLs will impact formulary design, cost sharing, rebating, and pharmacy and provider reimbursement, all of which can impact patients’ access to medications. Respondents highlighted potential costs to plans stemming from UPL implementation, which could put pressure on premiums and contractual arrangements with pharmacies and providers.
Background
As of March 2025, PDABs have been established in eight states (CO, MD, ME, MN, NH, NJ, OR, and WA) with the stated goal of reducing drug spending and patient out-of-pocket (OOP) costs. Of these eight PDABs, four have UPL-setting authority (CO, MD, MN, and WA),¹ and one (OR) has drafted a UPL report on potential effectuation mechanisms despite not having UPL authority. Some stakeholders have raised concerns that UPLs could increase supply chain costs, exacerbate shortages, or cause new patient access issues.
Health plans have a unique perspective regarding possible implications of UPLs on insurance coverage and other changes that may impact patient access. To gather these perspectives Avalere sought out plan perceptions on the impacts of UPLs and their preparedness for UPLs. Avalere updated and built on previous payer interviews (conducted in 2023 and released in 2024) with interviews and a survey of health plans.
Results
Increased Cost Sharing and Disruptions to Benefit Design Are Key Patient Access Considerations
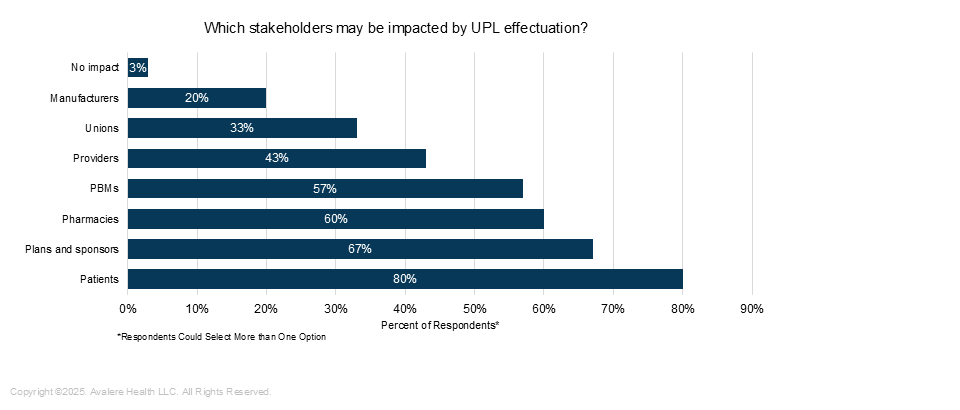
When asked which types of stakeholders may be most impacted by UPL effectuation, 80% of respondents selected patients—the highest out of all stakeholders listed (Figure 1).
Figure 1. Stakeholders That May Be Impacted by UPL Effectuation
 Respondents also answered questions about the potential patient impacts of UPLs on premiums, benefit design, and formularies. Overall, 60% of respondents expected changes to OOP costs and 50% expected increased copays or coinsurance on a UPL drug (Figure 2).
Respondents also answered questions about the potential patient impacts of UPLs on premiums, benefit design, and formularies. Overall, 60% of respondents expected changes to OOP costs and 50% expected increased copays or coinsurance on a UPL drug (Figure 2).
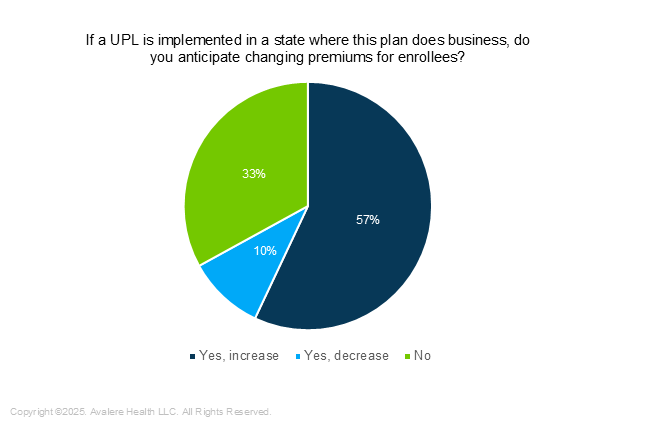
While just 50% of respondents expect that premiums would be disrupted by UPLs (Figure 2), 57% anticipate that premiums would increase if a UPL is implemented and 10% would expect lower premiums (Figure 3). Some interviewees noted the projected costs associated with implementation as a reason for higher premiums. A large minority (40%) of survey respondents expect that UPL implementation would result in higher administrative burden on plans, providers, pharmacies, or patients.
With respect to formularies and benefit design, payers expect the impact would vary based on which drugs are selected for a UPL. Most surveyed payers (83%) anticipate moderate or major disruption to formulary design.
Figure 2: Disruption Resulting From a UPL
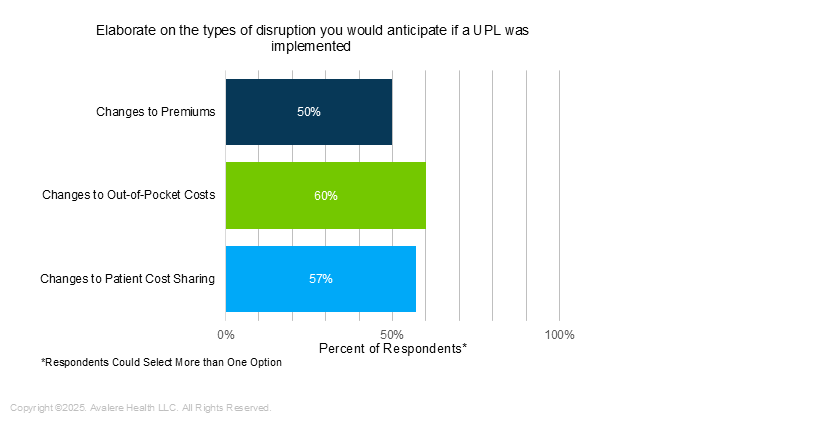
Figure 3. Changing Premiums as a Result of UPL Implementation
Respondents also considered whether payers would adjust tiering and formulary design in response to UPLs. Most (57%) survey respondents anticipate that both the UPL drug and its therapeutic alternatives would be implicated in formulary changes. When asked about other changes to benefit design, 50% of respondents said they foresee increased utilization management on UPL drugs.
Stocking Issues at Pharmacies and Gaps in Provider Reimbursement
Payers noted that setting UPLs could lead to changes for pharmacies, potentially impacting access for patients. Most (70%) respondents said that pharmacy reimbursement could decrease due to the implementation of UPLs. Moreover, 60% of respondents said those negative impacts on pharmacy reimbursement would decrease the likelihood of a pharmacy stocking a product with a UPL. Nearly three-fourths (73%) of payers surveyed agreed that this could lead to a shortage of the drug in the state with a UPL.
The survey also asked specifically about UPLs for physician-administered drugs and the impact on providers. Over half (57%) of respondents agreed that if a UPL were implemented, a provider would receive a UPL-based reimbursement that would likely be less than what the provider would otherwise be paid for that product. Nearly half (47%) of respondents indicated that patients could be responsible for making up the difference between acquisition cost and drug reimbursement, and 6% noted that providers may be at risk of not being able to make up that difference.
Diminished Rebates Could Contribute to Increased Premiums
Payers asserted that UPLs could impact manufacturer rebates for the UPL drugs and their therapeutic alternatives, with potential for discretionary rebates on these drugs to be reduced or eliminated.
For UPL drugs, 74% of respondents indicated that manufacturer rebates would either decrease or stay the same; 23% of respondents thought manufacturer rebates would increase.
For therapeutic alternatives to UPL drugs, 70% of respondents indicated that manufacturer rebates would either decrease or stay the same; 30% of respondents thought manufacturer rebates would increase.
Plans can use rebate dollars to reduce their premiums, so plans contemplating a reduction in those rebates—plus additional administrative costs noted above—could factor into their predictions of premium impact.
Conclusion
This research indicates that payers have concerns with the potential impact of UPLs on patients, pharmacists, providers, and their own financial positions. New issues have arisen since Avalere’s 2024 findings, including the potential for diminished access to UPL medicines, the need to update provider and pharmacy contracts, and formulary changes for UPL medicines and their therapeutic alternatives. Payers noted that these changes increase administrative burden, which could increase their costs.
As states continue to advance PDABs and UPLs, stakeholders should consider potential impacts on patient access, patient costs, provider reimbursement, plan impacts and benefit design, and manufacturer contracting.
Research Methodology
Avalere conducted interviews and a survey between January and February 2025. Both were double-blinded, with distinct respondent groups. Neither included individuals who were interviewed for Avalere’s similar work in 2023. Interviewees and survey participants included current and recent senior-level representatives of national and regional payers that had direct experience in prescription drug benefit design and had an ability to speak to an organization’s perception of UPLs and preparedness for implementation.
Avalere conducted six 30-45-minute interviews; interviewees represented 115.2 million total covered lives. The survey included 30 respondents, representing 476.3 million enrollees.²
The chart pack for this research can be found here.
Funding for this research was provided by the Partnership to Fight Chronic Disease. Avalere retained full editorial control.
¹ For this analysis, Avalere only included states that have passed legislation that establish PDABs that are required to conduct affordability reviews. For example, VTs Green Mountain Care Board has the option to conduct an affordability review of a set selection of drugs, but it is not a requirement.
² Surveyed payers did not identify their organization, thus there may be overlap of covered lives.





