Accelerated Approval of Drugs Gives Underserved Beneficiaries Access
Summary
A new Avalere analysis shows the Accelerated Approval pathway provides access to underserved Medicare beneficiaries across a range of conditions with unmet needs.The Accelerated Approval (AA) program provides opportunities for patients with unmet medical needs to access drugs on an earlier timeline than the traditional FDA approval process. The AA pathway allows the Food & Drug Administration (FDA) to approve drugs based on surrogate endpoints that are deemed likely to predict clinical benefit in the treatment population prior to completion of confirmatory trials. Manufacturers must conduct confirmatory trials after accelerated approval has been granted. Once the FDA has reviewed the additional trial data, accelerated approval may be withdrawn or “converted” to traditional approval.
In the coming years, continued manufacturer investment in the development of treatments for specific cancer types, rare diseases, and other conditions with unmet needs—such as Alzheimer’s disease—is likely to accelerate. This has led some policymakers to call for reform of the AA pathway with new obligations, such as time limits on manufacturers to conduct post-approval studies on AA drugs and requirements surrounding the completion of confirmatory trials. Proponents of AA, however, are concerned that some of the proposed changes on the pathway could limit access and have a detrimental impact on patients with unmet medical needs.
Researchers have sought to answer stakeholders’ questions by analyzing the relationship between the AA pathway and outcomes such as broader patient access and improvements in health status. However, it has been difficult to isolate the direct effects of the AA pathway given challenges associated with examining data for products that have received both traditional and accelerated approval for multiple indications.
Approach
To isolate utilization and access directly attributable to AA treatments among Medicare beneficiaries, Avalere conducted a multidimensional assessment of the AA pathway, focused only on the subset of products whose first indication was granted under AA through the first conversion date (i.e., drugs with market availability based solely on AA that would have otherwise not been accessible to patients).
The analysis included three components. The first examined discernible changes in utilization or access as measured by the total number of years that beneficiaries had relevant prescription fills as a direct result of the AA pathway. Second, Avalere examined if utilization and access patterns differed for racial minorities and those with lower income levels versus a relevant comparison group (non-Hispanic White beneficiaries and non-dually eligible beneficiaries, respectively). Finally, Avalere conducted a case study of a condition represented in this subset of drugs to analyze the relationship between earlier treatment access and the utilization of inpatient hospital services and emergency room (ED) visits.
Results
Patient Access Years Gained
Avalere estimated the total number of years that Medicare beneficiaries were able to access treatment by therapeutic area. As depicted in Table 1, drugs first approved under the AA pathway resulted in 257,639 beneficiary years of access to drugs, utilized by 182,352 unique beneficiaries.
| Therapeutic Area | Number of Drugs Evaluated | Beneficiary Years |
|---|---|---|
| Oncology | 54 | 176,905 |
| Neurology | 5 | 36 |
| Antidotes | 4 | 51,949 |
| Hematology | 2 | 1,384 |
| Cardiovascular | 1 | 16,862 |
| Bacterial Infection | 1 | 3,499 |
| Gastrointestinal | 1 | 5,491 |
| Genetic Disorders | 1 | 129 |
| Infectious Diseases | 1 | 134 |
| Parasitic Diseases | 1 | 12 |
| Women’s Health | 1 | 1,369 |
Access Among Subpopulations
Avalere also examined whether access to this subset of products was different for underserved populations (vs. relevant comparison groups of non-Hispanic White and non-dually eligible beneficiaries) based on demographic characteristics commonly associated with variations in access: race or ethnicity, income status, and geographic location (urban or rural). The assessment of access among vulnerable populations included 10 drugs in 7 therapeutic areas for which there was sufficient sample (with accelerated approvals 2011–2019). Eight of the ten drugs included are administered under Part D. Table 2 depicts access pattern differences by therapeutic area and beneficiary subgroup. Overall, lower-income beneficiaries had higher use of AA drugs compared to those with higher incomes.1 Additionally, in most cases non-White beneficiaries had equal or higher use of AA drugs compared to non-Hispanic White beneficiaries. In most cases, there were no differences in use between beneficiaries residing in rural or urban areas.
| Therapeutic Area | Low Income | Black/ African American | Hispanic | Rural vs. Urban |
|---|---|---|---|---|
| Oncology – Breast Cancer | ↑ | — | ↑ | — |
| Oncology – Immunotherapy | ↑ | ↑ | ↓ | ↑ |
| Oncology – Lymphoma | — | — | — | — |
| Oncology – Urothelial Cancer | ↑ | — | ↑ | — |
| Cardiovascular | ↓ | ↓ | ↓ | ↓ |
| Antidotes | ↑ | ↑ | — | — |
| Hematology | ↑ | ↑ | ↑ | — |
| Bacterial Infection | ↑ | — | — | — |
| Gastroenterology | ↑ | — | ↑ | — |
| Women’s Health | N/A | — | ↑ | — |
| ↑ | Higher use of AA drug compared to the reference sample (p<0.05) | ↓ | Lower use of AA drug compared to the reference sample (p<.0.05) | — | Statistically equivalent use |
Source: Avalere analysis of Medicare FFS Part A, B, and D claims.
Notes: N/A indicates insufficient sample size. Low income compares dually eligible beneficiaries to non-dually eligible beneficiaries. Black/African American compares Black/African American beneficiaries with non-Hispanic White beneficiaries, and Other Races/Ethnicities compares Other Races/Ethnicities to non-Hispanic White beneficiaries.
Case Study
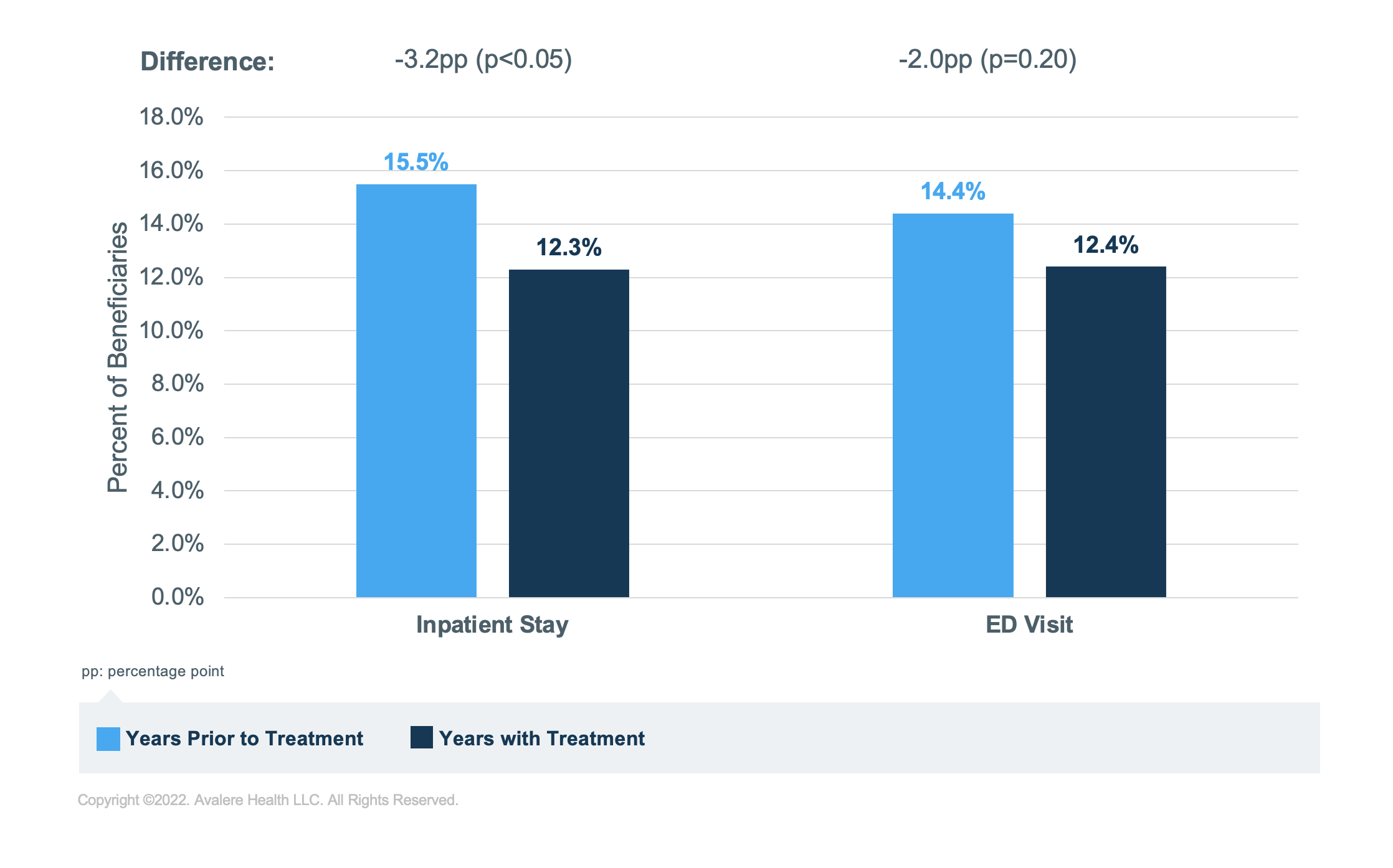
Avalere conducted a case study of a condition represented in this subset, primary biliary cirrhosis (PBC) to explore the relationship between treatment access and hospital utilization. Avalere selected PBC because there are no other approved drugs to treat the condition and approval was only for this condition. Among beneficiaries using the AA treatment, Avalere found that treatment was associated with a reduction in the rate of inpatient stays compared to the year prior to treatment initiation. The proportion of beneficiaries with PBC who had an inpatient stay decreased from 15.5% in the year prior to treatment to 12.3% after treatment (3.2 percentage point reduction, p<0.05). The difference in the proportion of beneficiaries with an ED visit was not statistically significant.
Implications
Overall, drugs with FDA-approved indications granted only via accelerated approval resulted in more than 250,000 beneficiary-years of access to drugs among Medicare enrollees with rare diseases and other conditions with unmet needs. These analyses were limited to Medicare beneficiaries, but many drugs approved via the AA pathway target pediatric conditions that were not reflected in this analysis.
Stakeholders may also consider the impact of these treatments on underserved subpopulations. In general, access to the AA drugs selected for this analysis was equal or higher among low-income beneficiaries (vs. non-dual eligible beneficiaries) and racial/ethnic minorities (vs. non-Hispanic White beneficiaries). Finally, Avalere’s case study showed that treatment with the examined AA drug was associated with a reduction in the utilization of inpatient hospital services. Taken together, these results underscore the key considerations that policymakers may further examine in any potential reform of the AA pathway.
Methodology
Fee-for-service (FFS) beneficiaries were identified using the 100% file of Medicare FFS Parts A, B, and D data, accessed via a research collaboration with Inovalon, Inc. and governed by a research-focused Centers for Medicare & Medicaid Services data-use agreement. Avalere selected drugs whose first indication was granted via AA between 2011 and 2022, from first date of approval through the first conversion date (i.e., the time on market solely related to AA) to isolate utilization and access directly attributable to AA treatments among Medicare beneficiaries. Drugs approved with indications granted under traditional approval were excluded. The RTI race code was used to identify beneficiary race or ethnicity.
Avalere conducted tests of differences in means to evaluate access and utilization of AA drugs among beneficiary subpopulations. Generalized linear models were used to evaluate the association between use of an AA treatment and utilization of hospital services, controlling for beneficiary demographics and year of diagnosis. Avalere considered outcomes to be statistically significant at p<0.05.
To learn more about the effect of AA pathways, connect with us.
Note
- Low income (dually eligible) beneficiaries had higher use of AA drugs within 7 of the 9 therapeutic areas with sufficient sample.





